
A potent insulator and arc quencher, SF6 gas is widely used in the world of transmission and distribution, as well as other applications. Find the answers to frequently asked questions about this gas’s attributes, risks, and best practices.
Sulfur hexafluoride (SF6) has extremely good electrical and thermal insulation properties. For that reason, it is widely used in médium-and high-voltage electrical power transmission and distribution (T&D). But it requires careful handling. Here are 15 FAQs about this gas, its chemical properties, and how to use it safely and efficiently.
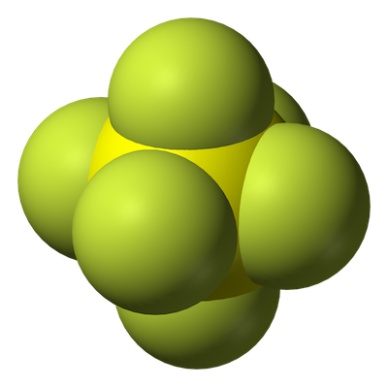
SF6 is made up of one sulfur atom surrounded by six fluorine atoms. Source: Wikipedia Commons
1. What is SF6 gas?
Sulfur hexafluoride is an octahedral (eight faces and six vertices) molecule. It consists of six fluorine atoms surrounding a sulfur atom. The chemical can occur in nature, but most SF6 gas is manufactured. Researchers in Paris were the first to synthesize SF6 in 1900. Large-scale production of this gas in the U.S. began in the 1950s.
2. What are the characteristics of SF6 gas?
SF6 is colorless, odorless, nonflammable, and nontoxic. It is virtually inert, which means it is stable and does not react with other chemicals under normal conditions.
3. Why is SF6 gas such a good dielectric medium?
First, dielectricity is the ability to transmit electricity without conductivity, and dielectric strength refers to how well something can withstand high voltage without breaking down. Electronegativity is the ability to capture free electrons, and fluorine is the most electronegative element known on earth. SF6 has six fluorine molecules that can break away from sulfur, capture electrons during an arc, and then return to its original state. The gas’s electronegativity and high dielectric strength are why it is such a popular insulating gas for use in power T&D equipment.
4. What is the importance of arc quenching in T&D equipment?
When switching or opening an electrical circuit that deals with more than 250 volts, as the contacts start to open, an arc forms between them. This arc can be more than 2,000°C (3,632°F), which is hot enough to melt and make the metal contacts stick to one another. When SF6 gas captures free electrons, it mitigates the effects of arcing.
5. Is SF6 gas toxic to humans?
SF6 is biologically inert and, therefore, is nontoxic to humans and animals. In fact, it is used in medical diagnostics as an ultrasound contrast agent and as a tamponade gas (to plug fluid flow) in ophthalmology.
6. What happens if someone breathes SF6 gas?
Not much. The gas itself is nontoxic and will not harm humans or animals. However, SF6 is much heavier than air and, therefore, can displace oxygen in the lungs and lead to asphyxiation if inhaled in extremely large quantities.
7. Is SF6 gas harmful to the environment?
This gas does not deplete the ozone layer or cause air pollution. It is, however, almost 24,000 times more effective than carbon dioxide (CO2) at trapping heat, making SF6 a very potent greenhouse gas. That’s why it’s so important to always monitor the SF6 level in electrical switchgears for leaks.
8. Can SF6 gas harm equipment?
As an inert gas, pure SF6 does not damage metals. However, if the gas has been contaminated with water, some of the byproducts generated during arc-quenching cycles are corrosive. See #13 for more information on water as an impurity.
9. What are the most common applications of SF6 gas in electrical power?
SF6 is routinely used as an electrical and heat insulator in medium- and high-voltage T&D applications. These include:
- Circuit breakers
- Switchgears
- Power, voltage, and current transformers
- Gas-insulated lines
- Capacitors
10. In what other industries are SF6 gas used?
Besides electrical T&D applications, SF6 is commonly found in radiological instruments, particle accelerators, equipment for semiconductor production, and magnesium and aluminum casting. It is also used as insulation in windows to dampen sound, and as a tracer gas in mining.
11. Why is SF6 the preferred insulator for breakers and switchgears?
SF6 is extremely effective at quenching the arc generated when switching or opening electrical circuits. It also has very high thermal conductivity, especially at high temperatures, and, therefore, dissipates heat much better than air, nitrogen, helium, or other gases. In addition, equipment insulated with SF6 has lower operating noise, does not emit hot gases, and require relatively little maintenance.
12. What’s the best way to check the integrity of an SF6 breaker?
Continuous online monitoring of SF6 density inside the chamber provides accurate and reliable information about gas conditions and possible leaks. Commercially available integrated gas transmitterscan measure gas pressure, humidity, and temperature, and use these parameters to calculate gas density.
13. Why is humidity a problem for SF6 breakers?
In a dry environment, SF6 can quickly break apart to capture free electrons, and then recombine. However, when moisture enters a breaker, the sulfur and fluorine molecules grab water’s oxygen molecules to form byproducts during high-temperature episodes. One result is that there is now less of the gas to act as insulator. The second problem is the byproducts themselves, many of which are toxic and corrosive. These byproducts include thionyl tetrafluoride (SOF4), thionyl fluoride (SOF2), sulfuryl fluoride (SO2F2), sulfur dioxide (SO2), and disulfur decafluoride (S2F10).
14. What can be done about humid and dirty SF6 gas?
Use equipment specifically designed to handle SF6 gas. WIKA has the gas handling equipment for all sizes of T&D equipment. The GPU-2000, one of our SF6 solutions, recovers and fills the gas in switchers and cleans the gas of impurities. The GAD-2000is able to dehydrate SF6 without having to take equipment offline.
15. Are there any regulations or standards regarding SF6 and its byproducts?
The IEC(International Electrotechnical Commission) has several SF6 standards in place including:
- IEC 603776 that defines SF6 quality and detection techniques for use in electrical equipment, and gives general information about the gas and its environmental effects.
- IEC 62271 regarding how to handle SF6 and its mixtures.
- IEC 60480 about effects on human health, handling, and disposal of SF6 byproducts.
- IEC 60068 on how to determine SF6 leakages.
Click here to download the white paper “GPU-2000: WIKA’s Optimal Solution for Comprehensive SF6 Gas Handling”



SF6 gas and its features and use in SF6 Breakers is extremely use full to engineers.
We are doing maintenance on SF6 breakers (345kV). Last time when the SF6 gas was being extracted the storage bottles froze up and the gas stopped flowing into the bottle. The contractor used a torch to free the clog. Two questions? is this safe way to free a clog. And the other question is what is the best way to heat the bottle or keep this from happening?
Hu Kelly, Thank you for your question. Our product manager, Leo, will be reaching out to you shortly if he has not already.
Hi Kelly, you can use an electric heating pad around the bottle valve to heat it while gas is flowing. There are some rubber-like pads which run on AC or DC. there are also resistive heating ropes with the same principle. Probably your SF6 provider has some readily available solutions.
Dear wika.us administrator, Thanks for the well written post!
Hi wika.us webmaster, You always provide key takeaways and summaries.
I would like to express my appreciation for the informative article titled “15 Most Frequently Asked Questions about SF6 Gas and SF6 Breakers” on the WIKA USA blog. This article provides valuable insights and answers to common questions regarding SF6 gas and SF6 breakers, shedding light on this important aspect of electrical engineering.
The article effectively addresses a wide range of questions related to SF6 gas and its application in circuit breakers. By providing clear and concise explanations, you offer readers a comprehensive understanding of SF6 gas properties, its advantages as an insulating medium, and its role in ensuring the reliable operation of circuit breakers.
I appreciate the article’s organization and the inclusion of frequently asked questions that cover various aspects of SF6 gas and breakers. From questions about the composition and properties of SF6 gas to inquiries about safety, maintenance, and environmental considerations, you cover a broad spectrum of topics, ensuring that readers have a well-rounded understanding of the subject matter.
Furthermore, the article’s commitment to safety is commendable. By addressing safety precautions, leak detection methods, and the importance of proper handling and disposal of SF6 gas, you prioritize the well-being of professionals working with SF6 gas and underscore the significance of responsible and environmentally conscious practices.
To further enhance the article’s impact, it might be beneficial to provide additional resources or references for readers who wish to delve deeper into the topic. This could involve linking to relevant industry standards, research papers, or case studies that explore specific applications or advancements in SF6 gas technology.
Additionally, incorporating real-life examples or practical tips based on industry experiences could add further value to the article. Sharing insights gained from working with SF6 gas and breakers in different contexts would provide readers with a more nuanced understanding of the challenges and best practices associated with their usage.
Overall, “15 Most Frequently Asked Questions about SF6 Gas and SF6 Breakers” is a valuable resource for individuals seeking to expand their knowledge about SF6 gas and its applications in circuit breakers. By addressing common questions, emphasizing safety considerations, and providing comprehensive explanations, you contribute to the understanding and awareness of this important aspect of electrical engineering.
Hey Jim, thank you so much for your comment. We will look into the things that you addressed and we appreciate your input.
It’s a high GWP product causing damage to the environment. Do we have any greener solution in place of SF6.?
Why can’t nitrogen be explored as an option.?
Hi Mahesh. Your question was passed along to our product expert. He should be reaching out to you shortly if he has not done so already.