Typical consumers think of natural gas as just a fuel that cooks their food and heats their homes. But for the chemical and petrochemical industries, it is both the fuel and the feedstock used in the production of hydrogen, ammonia, syngas, liquid natural gas (LNG), and the compounds necessary to make plastics.
Methane (CH4) is the main and most valuable component of natural gas. For all usages, natural gas needs to be processed first to get rid of unwanted compounds:
- Water vapor
- Carbon dioxide (CO2)
- Sulfur compounds: hydrogen sulfide (H2S), carbonyl sulfide (CS2), carbon disulfide (CO2), mercaptans (RSH), etc.
The most common and harmful contaminants in untreated natural gas are carbon dioxide and hydrogen sulfide. The former is a major factor in global warming. The latter, also called hydrosulfuric acid, is an irritant at low levels and deadly in higher concentrations. These acid gases and other compounds also get in the way of producing hydrogen, ammonia, methanol, etc.
Amine Units: Technology for Treating Natural Gas
Processing plants remove as much of these acid gases as possible before piping the natural gas to consumers or production units, and one of the best technologies for treating natural gas is the amine system. It gets its name from the amine solution used in the process. This liquid with several alkylamine is a solvent that captures CO2 and H2S as it passes through raw natural gas. Carbon removal and gas treating, also called “sweetening” as it removes the rotten-egg odor from sour gas, are a necessary step nits in natural gas processing.
How an Amine Unit Works
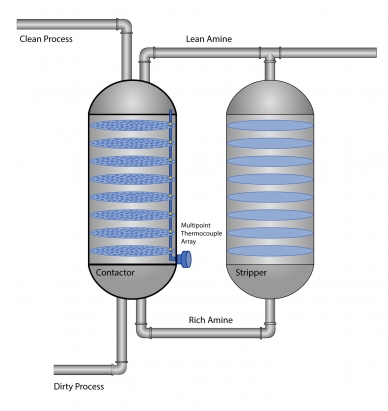
An amine unit consists of two main parts for two separate but interconnected processes: absorption and stripping. Here is a cursory overview of how a typical amine unit works.
The absorption process
- The sour gas (dirty process) enters the amine column, also called the contactor column. At the same time, lean amine (without acid gases) is pumped in from the top and rains down onto a series of perforated contactor trays.
- The dirty process gas naturally flows upward through the trays. As the amine solution on each tray makes contact with the rising gas, it absorbs any carbon dioxide and hydrogen sulfide molecules that it encounters.
- The higher the sour gas rises, the sweeter it becomes. At the top of the column, the treated gas is ready for use or further processing.
- The amine solution, now saturated with acid gases (rich amine), collects at the bottom of the column.
The stripping process
- A level controller at the bottom of the amine column detects the level of rich amine. When it reaches a predetermined height, this cooler rich amine is pumped into a heat exchanger and cross heated with hot lean amine.
- Now hot, the rich amine enters the stripping column. Steam from the regenerator separates the acid gases from the rich amine, thereby regenerating the solvent. A filtration system further strips away any residual contaminates. In a separate process, the acid gas is cooled, condensed, and removed from the stripping column.
- The hot amine solution, now stripped of acid gases, is cooled and pumped back to the amine contactor column, where the process begins again.
The Importance of Temperature Monitoring and Control in Amine Units
Amine units require precise controls of pressure, level, and temperature, but especially the latter. Poor temperature control presents several challenging issues:
- Condensation and foaming. The cooler the lean amine, the slower it flows and the better it absorbs acid gases. However, if the solvent is cooler than the dirty process gas, the hydrocarbons could condense and possibly foam. Therefore, the lean amine needs to be just slightly hotter than the gas inlet temperature for thorough gas treating.
- Poor absorption. Conversely, the hotter the lean amine, the faster it flows. An increased flow rate means it doesn’t spend enough time with the dirty process gas to react with (absorb) the CO2 and H2S, leading to poor product quality.
- Carryover. Most of the reaction should occur at the bottom third of the contactor column. But if the dirty process is too hot as it enters the tower, it will rise too quickly and come into contact with the lean amine too near the clean process outlet. This reaction results in foaming, which then carries some of the solvent and dirty process over into the clean process gas as it exits the column, leading to poor product quality as well as amine loss.
- Corrosion. Amines naturally inhibit corrosion. However, in areas with temperatures higher than 266–275°F (130–135°C), acid gases can break out and attack metal surfaces.
- Inefficient amine filtering. As the amine solution is constantly stripped and reused, it needs to be as lean as possible in order to capture as many contaminants as possible. One of the ways to maintain a high-quality solvent is to pass the solution through a filtering system. For maximum efficiency in this step, the operating temperature must be higher than 122°F (50°C).
An optimized process of treating gas improves overall efficiency. However, suboptimal processes in an amine unit can negatively affect the operations and profitability of the entire plant.
- Frozen storage tanks. Cryogenic processes are found in natural gas plants and other process industries to recover and purify products that are gaseous at ambient temperature and pressure. If too much carbon dioxide remains in the treated gas, CO2 molecules will freeze at a higher temperature (−2°F/−78.5°C) than natural gas, which liquifies at approximately −260°F (−162°C). When this occurs, frozen CO2 (dry ice) will plug storage equipment and cause other operating problems. In case of an outage, a hydrogen plant of 100,000 Nm3/h will lose about $100,000 per day in the value of the hydrogen alone. This figure does not include the other processes that are disrupted, such as ammonia or methanol production.
- Unnecessary energy expenditure. As rich amine must be heated for optimal acid-gas separation in the stripper, a too-low temperature as it exits the contactor column would require additional energy before the next step of the process.
- Unnecessarily low throughputs. When plant operators do not have a complete temperature profile of the contactor column, they will be cautious and reduce the inlet process gas flow to minimize carry over. A lower throughput is called for in certain situations, but unnecessary capacity reductions are costly. For example, a 5% capacity reduction results in lost opportunity of $2.2 million per year.
This two-part series continues with “How Multipoint Thermocouple Arrays in Amine Units Increase Production and Prevent Failures.”