WIKA USA recently had the privilege of partnering with Chevron to custom-build multipoint thermocouple arrays for their refineries’ contactor columns. A chemical specialist from WIKA and a process control engineers from Chevron jointly presented the following paper at the CRU Sulphur + Sulphuric Acid 2023 conference in New Orleans.
Abstract
Amine units are systems that treat, or “sweeten,” raw natural gas by capturing and removing unwanted compounds, primarily hydrogen sulfide (H2S) and carbon dioxide (CO2). An optimized gas sweetening process is essential for a plant’s overall efficiency and profitability.
Temperature monitoring and control are necessary for operating amine units, as that is how refineries and chemical plants can prevent some of the major issues seen in amine contactor columns. Reliable temperature information and a complete temperature profile help operators optimize their units’ performance by pinpointing where most of the heat of absorption is occurring in the column. Using this information, they can minimize and even avoid foaming, carryover, corrosion, inefficient amine filtering, and other problems.
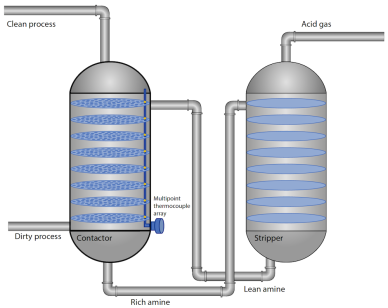
A typical contactor column has around 20 trays stacked at an equal distance from each other. A single thermocouple placed somewhere along its height provides but one data point, whereas a multipoint temperature system offers a complete temperature profile of the column. WIKA designed, manufactured and installed multipoint thermocouple assemblies specifically for two of Chevron’s refineries. This temperature measurement solution helped the company maximize its amine units’ efficiency, which resulted in significant insights that allowed for proactive responses to avoid operational upsets, as well as opportunities to improve efficiency through optimized operation.
Introduction and Process Overview
Sour gas contains methane, acid gases (like H2S and CO2), and, commonly, low levels of hydrogen, ethane, and heavier hydrocarbons as well. Before raw natural gas can be used as either fuel or as feedstock for the production of hydrogen, syngas, and other valuable products in a refinery, it must first be processed to remove contaminants:
- Carbon dioxide (CO2)
- Sulfur compounds, such as hydrogen sulfide (H2S), carbonyl sulfide (CS2), carbon disulfide (CO2), mercaptans (RSH)
- Water vapor (H2O)
The most common and harmful contaminants in sour gas are carbon dioxide and hydrogen sulfide. The former is a major factor in global warming. The latter, also called hydrosulfuric acid, is an irritant at low levels and deadly in higher concentrations. These acid gases and other unwanted compounds also hinder the production of hydrogen, ammonia, methanol, etc.
One of the most common technologies for removing contaminants from dirty process gas is the amine unit. It uses an amine solution – a solvent with several alkylamines, like diethanolamine (DEA) – to capture CO2 and H2S as the liquid passes through sour gas. One study found that maximum CO2 removal is achieved at a lean amine concentration of 30 wt%, a temperature of 50°C (104°F), and a circulation rate of 260 m3h−1 (Darani et al., 2021). The researchers found that the impact of temperature on absorption was comparable to that of the circulation rate, contributing 20.3% to the overall results.
Amine units are critical to the operations of refineries and chemical plants. If a unit fails due to condensation and foaming, corrosion, or another issue, the entire facility’s productivity and safety are at risk.
How an Amine Unit Works
An amine unit consists of two main parts for two separate but interconnected processes: absorption and stripping.
The absorption process
- Dirty process gas enters from the bottom of the contactor column (absorber) and naturally flows up through a series of perforated contactor trays (or packing, for some applications). At the same time, lean amine (solvent with very low levels of acid gases) is pumped in from the top and rains down through the trays/packing.
- As the amine solution on each tray makes contact with the rising dirty process gas, it absorbs the carbon dioxide and hydrogen sulfide molecules that it encounters.
- Consequently, the higher the gas rises, the sweeter it becomes. At the top of the column, the gas is now treated and ready for use or further processing.
- The amine solution, now containing higher levels of acid gases (rich amine), collects at the bottom of the column before it is transferred to the regenerator/stripper.
The stripping process
- The volume of relatively cool rich amine in the bottom of the contactor is pumped through a heat exchanger (not shown) and cross-heated with hot lean amine.
- Now hot, the rich amine enters the stripping column from near the top. Steam from the regenerator separates the acid gases from the rich amine, thereby regenerating it. A filtration system – solid filters and/or fixed carbon beds – further removes any residual contaminates from the solvent. In a separate process, the acid gas is cooled, condensed, and removed from the stripping column.
- The hot amine solution, now stripped of acid gases (lean amine), is cooled and pumped back into the amine contactor column, where the process begins again.
Current challenges in amine gas treating
An optimized process of treating sour gas improves a plant’s overall efficiency. Similarly, suboptimal processes in an amine unit can negatively affect the operations and profitability of the entire plant.
- Unnecessary energy expenditure. As rich amine must be heated for optimal acid-gas separation in the stripper, a too-low temperature as it exits the contactor column would require additional energy before the next step of the process.
- Unnecessarily low throughputs. When plant operators do not have a complete temperature profile of the contactor column, they will be cautious and reduce the inlet process gas flow to minimize carry over. A lower throughput is called for in certain situations, but unnecessary capacity reductions are costly.
Measuring Temperature in Amine Columns: Best Practices
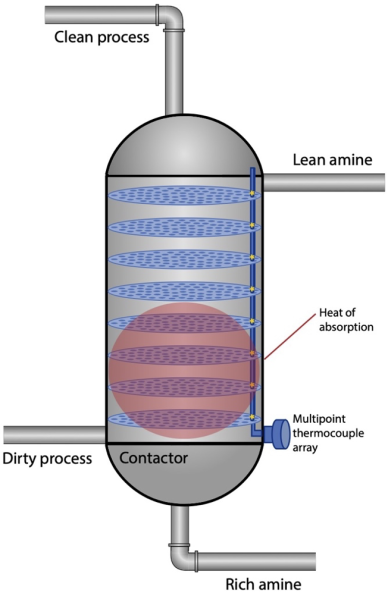
A typical amine contactor column has 20 to 25 trays stacked equal distances apart, or equivalent height of random packing. Sour gas enters the amine column at a relatively low temperature and is exposed to the lean amine, whose temperature is slightly higher, on each tray. When the two media meet, the process is exothermic, and the dirty process gas will increase in temperature.
Ideally, the bulk of the acid-gas capture – detectable by a temperature bulge – takes place low in the column so that there is adequate opportunity for refining the gas in the remaining height. Maintaining this optimal position will minimize the risk of hydrocarbon foaming or the breakthrough of acid gas into the treated gas.

Fig. 2: Simulated temperature and H2S concentration from a typical sour gas absorber in a refinery (click to enlarge)
Commonly available process simulation packages allow for estimations of temperature and H2S/CO2 content of the gas stream as it travels up the tower. Figure 2 shows extracted temperature and H2S concentration from a typical refinery sour gas absorber (same data on both but different scales for top x-axis). A smooth approach to temperature equilibrium with the incoming amine temperature, as shown by the solid trend with an adequately high amine circulation rate, can be a good indication for ensuring equilibrium in gas treating by the time the treated gas exits the top of the contactor, as little or no further absorption of H2S or CO2 is needed in the top “polishing” section. The extent to which H2S is removed can be highly nonlinear with changes to amine circulation rate. The dashed lines show how quickly a ~3% change in circulation rate can turn into a treated gas exceedance when the unit is close to a breakthrough point.
Some amine columns have no internal temperature measurement or just one thermocouple installed along its length – usually low. This single indication is better than not having any temperature sensors at all, but a multipoint thermocouple array provides a complete and detailed temperature profile, letting operators know precisely where most of the heat of absorption is taking place (see Figure 3).
With this information, operators can adjust the flow rate of the dirty process gas to minimize the unit’s energy consumption, maximize its efficiency, and avoid the issues mentioned above. While exact treated gas concentrations throughout the contactor column are not very practical in this application, having multiple temperature indications can give tremendous insight into how well the gas is being treated and, thus, help operators identify opportunities for unit optimization.
Multipoint Temperature System for Amine Contactors
Operating facilities, in their efforts to become more cost-efficient and to minimize carbon emissions, are always seeking ways to reduce energy consumption. One of those ways is to improve the efficiency of its amine units.
The most energy-intensive process in an amine unit is stripping, when the solvent is heated and regenerated by removing the CO2 and H2S that it captured in the contactor column. A majority of the heat input for the regeneration process is a function more so of the heat required to increase the temperature of the circulating amine from amine exit to stripper operating temperature (sensible heat) than the amount of acid gases that the solvent has absorbed, so this regeneration process is most efficient if solvent circulation rate is optimized for the contactor column. This is accomplished by reducing total amine circulation when possible. Knowing where the heat of absorption is taking place, and modifying amine circulation as necessary to adjust its location, allows for real-time monitoring and optimization of the amine unit.
This two-part series continues with an in-depth look into our custom-made multipoint thermocouple array for Chevron’s amine unit’s contactor column.