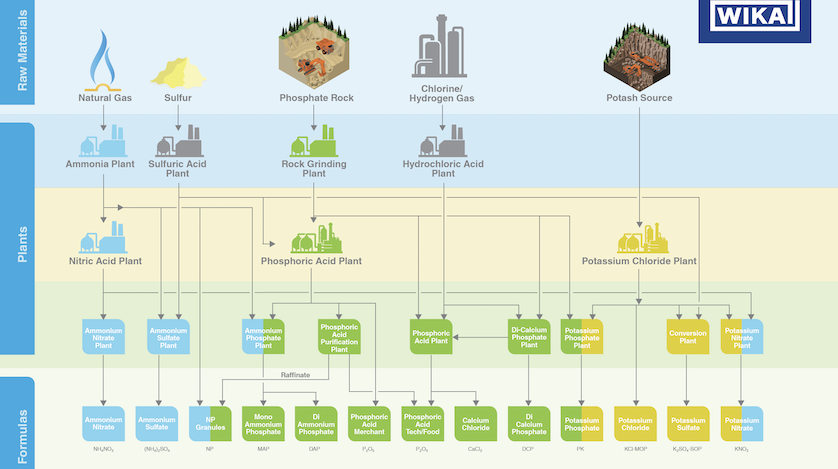
The fertilizer industry converts raw materials into three main types of fertilizers: nitrogen (ammonia), phosphorus, and potassium. These various applications call for chemical processes with precise control and accurate monitoring of temperature, pressure, level, and flow.
The global population is projected to reach 10.9 billion by the end of this century. This pressure pushes growers to increase crop yields, which is a more eco-friendly approach to food production than deforestation. Add in a demand for biofuels, much of which currently come from crops, and it becomes clear that the need for chemical fertilizers is greater than ever.
Each year, the fertilizer industry transforms millions of tons of raw materials – air, natural gas, and mined ores – into products that supply plants with three essential nutrients: nitrogen, phosphorus, and potassium.
Three Main Classes of Fertilizers
1. Nitrate fertilizers
Nitrogen-based fertilizers, the largest and most important group, take several steps to make. The raw materials are the nitrogen in air and the hydrogen from natural gas/methane (CH4). When mixed under high temperature and pressure, the resulting product is ammonia (NH3). This intermediate product is oxidized to produce nitric acid (HNO3), which results in the mineral fertilizers of ammonium nitrate (AN) and, when mixed with CO2, urea. A third type of nitrogen-based fertilizer is urea ammonium nitrate (UAN), made by mixing AN, urea, and water.
2. Phosphorus fertilizers
This group of fertilizers come from phosphate rock, a mined ore. When phosphate concentrate is treated with sulfuric acid (H2SO4), it becomes either single superphosphate (SSP) or phosphoric acid. This acid is mixed with ammonia to produce monoammonium phosphate (MAP) or diammonium phosphate (DAP). The fertilizer triple superphosphate (TSP) can be made by concentrating phosphoric acid or further concentrating phosphate.
3. Potassium fertilizers
This fertilizer is also derived from mined ore: potash rock, an amalgam of potassium carbonate and potassium salts. The process for making potassium fertilizers begins with concentrating potash and then treating it to make a potassium chloride solution. From this solution comes muriate of potash (MOP), potassium nitrate (KN) when mixed with nitric acid, and sulfate of potash (SOP) when mixed with sulfuric acid.
Process Challenges in the Fertilizer Industry
Throughout the various processes, fertilizer production requires the precise control and accurate monitoring of high temperatures and pressures, as well as the level and flow of feedstocks and catalysts. Moreover, as fertilizers and catalysts are caustic, measuring instruments must be able to withstand harsh media and conditions.
Each process has its specific challenges. At the heart of fertilizer production is the demand for safety, reliability, and efficiency.
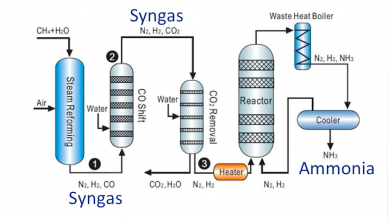
Nitrogen-based fertilizers: syngas and ammonia process from natural gas
Challenges in ammonia production
Most ammonia manufacturers today use the Haber-Bosch process, which calls for extremely high pressures, moderately high temperatures, and catalysts to cause a chemical reaction. One of the biggest challenges is finding the right balance between pressure and temperature in order to increase the kinetics of the nitrogen and hydrogen reaction to the target ammonia conversion.
An increasingly popular method of obtaining ammonia, due to its renewable nature, is electrochemical processing using electrolysis. Depending on the type of electrolyte used, this process also involves medium-to-high temperature and pressure conditions for ammonia conversion.
Challenges in phosphoric acid production
Phosphoric acid is manufactured using two processes. The “wet” process yields an acid of lower purity. However, this weaker acid is commonly used as fertilizer without requiring further processing or undergoing a thermal reaction to produce a higher-purity acid.
In the wet process, concentrated sulfuric acid is added to phosphate rock in a series of well-stirred reactors, followed by filtration to separate out byproducts. Finally, evaporation increases the phosphoric acid concentration to produce a commercial-grade fertilizer. One of the wet process’s biggest challenges is the highly corrosive nature of the sulfuric acid used in the reaction.
The other method of manufacturing phosphoric acid is the thermal process. Phosphorus is burned in air at about 1,800°K to 3,000°K, followed by direct hydration via steam to obtain phosphoric acid of approximately 85% concentration. Phosphates, the salts of phosphoric acid, can be further processed in combination with ammonia to obtain different fertilizer compounds. One of the thermal process’s biggest challenges is the extremely corrosive conditions created during production.
Challenges in potassium chloride production
Potash fertilizers are obtained from mined ore. First, potassium ores are injected with hot water to create the brine used as feedstock. Next in the process is an evaporation stage using a thermo-compressor, followed by a series of progressively lower temperatures and pressure crystallizations to recover potassium chloride, the key intermediate product. One of the biggest challenges in KCl production is to maintain accurate pressure and temperature profiles for system efficiency. Water flow is also an important parameter to control, as this ensures product purity and grain size.
Fertilizer producers are increasing efforts to improve performance in order to become a safer, more reliable, and more environmentally friendly industry. This goal will be achieved only with innovative, high-performance instrumentation for process control.


Hello wika.us owner, Your posts are always well-received by the community.
Thank you for such an interesting and relatively unknown (by the average consumer of fertilizers, etc.) discussion of the background processes used to create products in daily use. More discussion of short and long-term environmental consequences, both in the creation and the application, of the products would be appreciated, as well as more environmentally friendly alternatives, if any.